Aims and scope
Translational and Regulatory Sciences (TRS)
Purpose of TRS
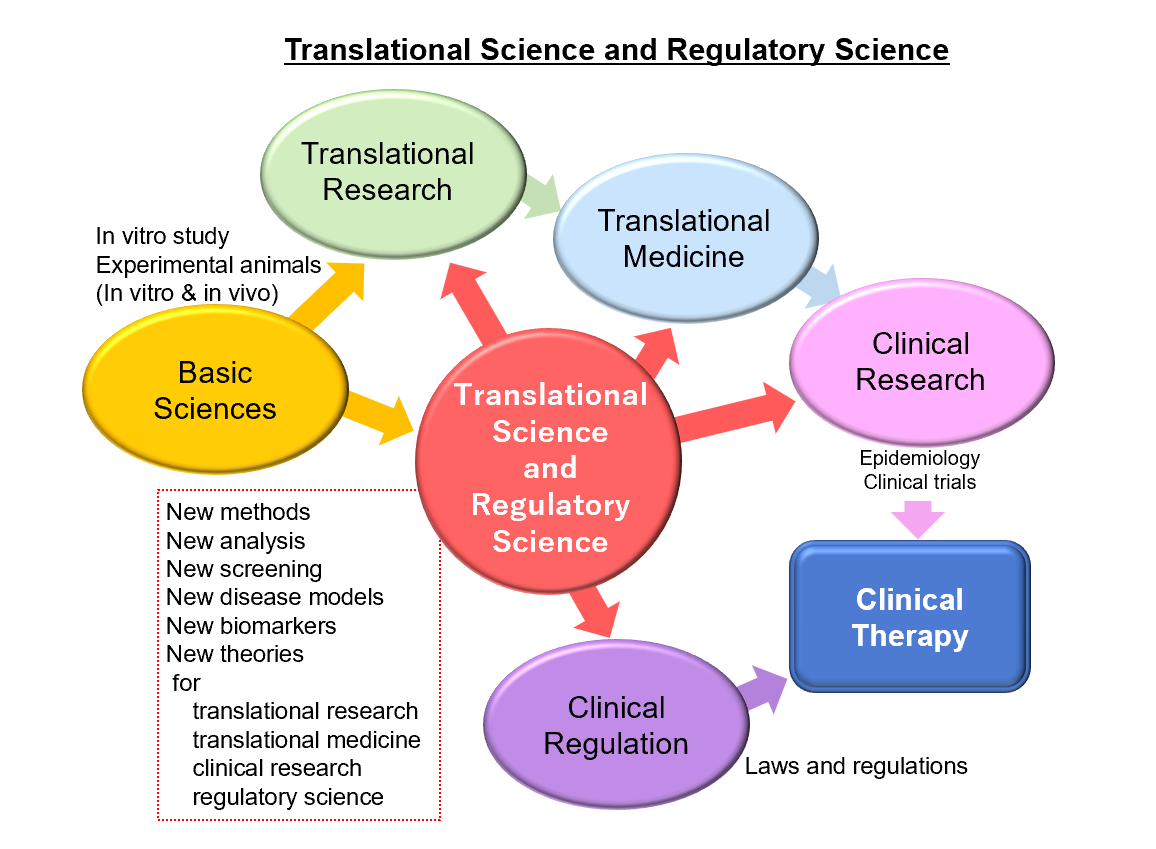
Translational science is a field of biomedical sciences. Translational science is different from translational research and translational medicine. Translational research is wide and encompasses everything between non-clinical and developmental research, and aims to establish the results obtained in basic research in the academe as new medical technology/medicine that can be used clinically. In translational medicine, research results obtained in translational research are applied in clinical trials and lead to the actual clinical setting. Meanwhile, translational science is the underlying research of translational research, and it refers to basic research that has the potential to advance to translational research in the future and that contributes to the development of regulatory science.
Regulatory science in the development of medicine indicates the basic and applied sciences that refer to the establishment of test studies to ensure the quality, safety, and effectiveness of pharmaceuticals and medical devices, and to establish evaluations, prediction, and judgment. Regulatory science is a scientific field that is also required for the inspection, safety evaluation, manufacturing control, and quality control of pharmaceuticals and medical devices, as well as for inspection and internal audit, such as Good Manufacturing Practice (GMP), Good Laboratory Practice (GLP), and Good Clinical Practice (GCP).
Translational science and regulatory science have quickly evolved over the last decade, and are both interdisciplinary and multidisciplinary, depending upon a large number of basic and applied scientific disciplines. Translational science and regulatory science are mutually involved in a complex manner. Therefore, both have to progress scientifically with respect to each other.
Translational and Regulatory Sciences (TRS) creates a new platform for launching new ideas, not only for industry and regulatory authorities but also by members of the academe who want to contribute to the better use of their research activities within medical aspects. Thus, TRS significantly impacts the abilities of these communities to bring new medicines and medical devices to patients in need.
The expected effect of TRS is that industry, academic, and government experts will harmonize their efforts on equal footing and will promote academic progress and dissemination regarding the regulatory science of pharmaceuticals, medical devices, and related aspects using this journal.
Spectrum of TRS
-
Original article (Full paper, Short communication)The journal adopts a peer-review system and aims for publishing high-quality research.The translational and regulatory science (TRS) spectrum represents each stage of research along the path from the biological basis of health and disease to interventions, including core technologies and clinical innovation. Through these stages, innovative methods and approaches that improve and accelerate the development of new pharmaceuticals and medical devices for translational and regulatory sciences are created and tested. TRS also accepts papers on toxicity evaluation and basic data necessary for new drug applications.
-
ReviewPost type: adopts a peer-review system Request type: by the planning and recommendation of the editorial committeeA comprehensive review of basic research, clinical applications, or regulatory science in an area of research, including your own research and integrating it with other published research.
-
Overview(Commentary)An over-viewed article outlining the current status and future of drug discovery development and its regulatory science based on the published literature on a particular area of research.
-
NewsIntroduces highly promising and epoch-making practical cases and new drugsIntroduces new regulationsIntroduces new facilities for drug discovery of the world
-
PlatformThe journal invites opinion from both the translational science and regulatory science sides regarding problems and direction in current and future drug discovery research worldwide.
Scope of TRS
Translational Sciences Section
- Establishment of new technology for drug discovery and drug toxicity evaluation: assay system(including high throughput screening (HTS), antibody, cell line, diagnostic marker, drug delivery system, target identification and validation, de-risking therapeutic development, cell therapy, regenerative medicine, gene therapy, and so on
- Establishment of new animal models of human diseases
- Identification of new target molecules and/or signaling in human diseases
- Translational clinical sciences in experimental small animals, from dogs and monkeys to humans
- New technology, method, and/or theory for analytical chemistry, early- or late-stage drug repurposing, compound library management, collaboration and partnerships, data transparency and sharing, patient and contact registries
Regulatory Sciences Section
- Evaluation of efficacy of new drugs before approval
- Evaluation of toxicity of new target molecules
- Proof of safety of new target compounds, antibodies, drug delivery systems
- New method to identify toxicity using new technology
- New concepts in benefit/risk assessment, submission and approval strategies, patient involvement, and ethical aspects
- New standards and approaches to facilitate sound and transparent regulatory decision-making regarding drugs and medical devices
- New standards, concepts, proposals, and/or regulations for gene therapy, cell therapy, and regenerative medicine

